The following post is a deeper look into a potential theory I have been researching which links PAS to progesterone, this is of course just a theory based upon various studies I have looked at and read. I recommend reading the whole thing as it would be great to get some feedback. If you don’t have time I will try and briefly summarise it here but I recommend you read the full thing:
It is very possible that PAS and sexual dysfunction relate in some way to dysfunctional dopamine signalling in regions of the brain such as the reward system. Dopamine signalling in these regions play a significant role in motivation, reward seeking behavior, pleasure and sexual function. The questions is how has this dysfunction arisen? Many have blamed androgens and problems with androgen signalling such as changes in androgen receptor expression etc. which could be correct but it is possible these issues also relate to progesterone.
Progesterone plays a significant role in sexual function in both males and females. Progesterone also has a significant effect on dopaminergic signalling, progesterone receptors are expressed on dopaminergic neurons in the brains reward system, where they can alter the ability of dopaminergic neurons to function. A study of human glial cells found that retinoic acid (a metabolite of isotretinoin) specifically increased neurosteroids pregnenolone and progesterone, in addition there was a significant increase in enzyme expression ( 4.9, 5 and 50 fold respectively ) which seems to favour progesterone production. Glial cells are the most abundant cell type in the brain, they surround neurons and are known to release active metabolites which affect neuronal function. Given this study was conducted in human glial cells it offers very strong evidence for what may occur in the human brain during isotretinoin treatment.
In sufficient amounts, progesterone can be a potent inhibitor of 5AR activity, its metabolites also antagonise the androgen receptor and studies have found progesterone can reduce androgen receptor expression in certain parts of the brain. Notwithstanding, as mentioned before, progesterone can also directly effect the dopamine signalling in brain regions such as the striatum which is crucial to the brains reward system. Given these factors, could progesterone signalling be responsible for PAS?
Progesterone also helps to explain anomalies such as low blood results for FSH and LH reported by people with PAS, as well as a reason for why drugs such as L-Dopa which act directly on the dopaminergic system don’t seem to have an effect. It also explains random reports that the effect of dopaminergic drugs such as cannabis, adderall and ritalin seem to have changed before and after isotretinoin.
Given this, I think there is good reason to look into the possibility that PAS could be related to progesterone and thus look at potential protocols to deal with this. I haven’t seen many PAS people try protocols which specifically deal with progesterone as this is often not considered as important.
Flynn - The progesterone theory of PAS
PAS and most major issues associated with sexual dysfunction such as low libido are likely caused by changes in dopaminergic signalling in the brain, particularly the dopaminergic reward system. Androgens for example ultimately act on androgen receptors in the brain to stimulate dopaminergic signalling associated with reward seeking and reward processing pathways linked to sexual function. Therefore, issues with androgen signalling ultimately lead to issues with dopamine signalling and thus potentially to sexual dysfunction. However, what is not clear is whether these changes in dopamine signalling are directly or indirectly affected by isotretinoin. For example, if isotretinoin has affected androgen receptor expression or androgen signalling, this would be an indirect effect on dopaminergic signalling.
Here I outline what I consider to be a strong case for the involvement of progesterone in the development of PAS.
Progesterone and progestogens (natural or synthetic steroid hormones which interact with the progesterone receptor) are known to have an effect on sexual function in males and females. An interesting phenomena relates to the steroid deca-durabolin (nandrolone decanoate) which is used by body builders and has progestin activity. The use of this steroid can lead to a condition nicknamed “deca dick”, in which users develop sexual problems such as loss of libido, erectile dysfunction, genital numbness, loss of interest in sex and a loss of pleasure from orgasm as well as mental effects such as loss of motivation, emotional flatness and apathy. It is thought that these effects can be avoided by taking testosterone alongside deca however there are numerous online accounts of people who report developing “deca dick” despite using testosterone. Interestingly, users sometimes report sexual problems such as low libido persisting long after stopping Deca despite normal androgen levels (1). One user reported these problems persisting for over a year and included emotional flatness and him thinking that “the reward function in my brain has fucked,”. Another user claimed he had felt no improvement after 5 years. Another such steroid with similarly reported effects is Trenbolone also known as Tren. Both of these steroids have activity at the progesterone receptor (PR). Deca durabolin is regarded as a potent progestogen and binds the PR with 22% of the affinity of progesterone. Trenebolone also binds with high affinity to the PR. Coincidentally, of all steroids which are used and abused, the only steroids with long term sexual side effects such as these seem to all be progestogens.
What makes these cases so interesting is that they are examples of persistent sexual dysfunction (loss of interest in sex, low libido, loss of morning erections etc.) as well as loss of motivation, low mood, apathy and feelings of being disconnected from your normal self in spite of normal androgen and hormone levels, as is the case in PAS. Another progestogen called depo provera is also capable of reducing sex drive/libido and has even been suggested for use in sex offenders to reduce sex drive, this shows the powerful effect progestogens can have on sexual function (2).
In the case of “deca dick”, numerous sufferers report these symptoms lasting for years after stopping the drug with no sign of improvement, some users claim no improvement after 4 or 5 years (3). This is a very long time to experience these kinds of severe side effects after taking a steroid. Again, these kinds of time periods suggest that a similar mechanism could be involved in PAS given the persistent nature of these effects. It also raises the possibility that progesterone signalling could be become dysfunctional or cause an imbalance in normal brain signalling which needs to be corrected before side effects subside.
Interestingly, some of these individuals with “deca dick” have tried things like L-Dopa, L-Tyrosine and Cabergoline to enhance dopamine signalling but gained no effect or benefit. This is also significant as progesterone receptors have been found to be expressed on many dopaminergic neurons in the brains reward system and their function is crucial to the activity of these neurons and dopamine signalling. Thus in theory, issues with progesterone signalling could alter dopaminergic signalling and thus even direct attempts to alter dopamine signalling through the use of L-Dopa or drugs would be ineffective without first dealing with progesterone. So what about isotretinoin and progesterone?
There are several studies linking retinoic acid to changes in progesterone, one study found that retinoic acid (metabolite of isotretinoin) significantly increased production and accumulation of progesterone in rat granulosa cells (4). Two other studies found that retinoic acid significantly reduced progesterone receptor expression in human breast cancer cells (5,6). The question is whether these same effects on progesterone receptor expression would occur in the brain or if this is specific to breast cancer cells. However the most interesting study was of a human glial cell lines which found that retinoic acid increased the production of pregnenolone and progesterone. This study found that retinoic acid increased the expression of cytochrome P450scc , steroid acute regulatory protein (StAR) and 3beta-hydroxysteroid dehydrogenase (3b-HSD) by 4.9, 5 and 50 fold respectively (7). These increases in enzyme expression are very significant, especially when you consider the steps of steroidogenesis that they catalyse.
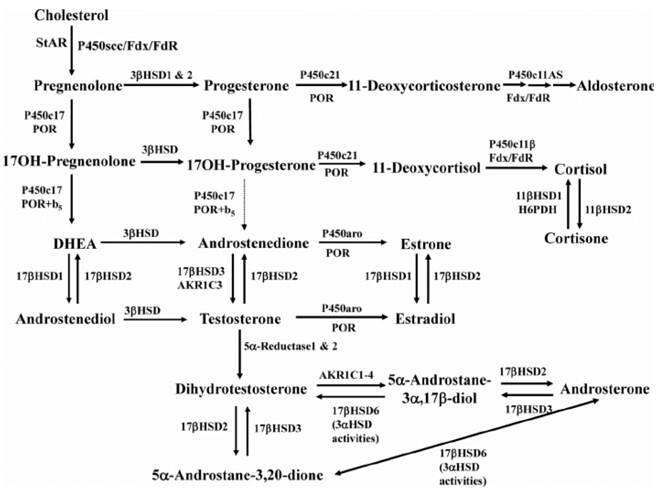
When we look at the steroidogenesis pathway, the enzymes whose expression is greatly increased by retinoic acid, P450scc, StAR and 3b-HSD, appear to favour the production of progesterone (please correct me if I am wrong on this). What is not completely clear is which type of 3b-HSD enzyme retinoic acid increased as type 1 and type 2 are involved in the conversion of pregnenolone to progesterone. Despite this, irrespective of these enzymes, the study itself noted that of all neursteroids, retinoic acid increased the de novo synthesis of pregnenolone and progesterone, this alone gives strong evidence to suggest that isotretinoin treatment can lead to significantly increased production of pregnenolone and progesterone over other neurosteroids in the brain by glial cells. It is important to note that glial cells are the most abundant cell type in the brain and they are known to release active metabolite which affect neuronal function. I am yet to see a study related to isotretinoin on a cell type which is more relatable to what may happen in the normal human brain during isotretinoin treatment.
Below is a diagram outlining the basic steps in a steroidogenesis pathway along with the specific enzymes involved at each step.
There are several things to note about progesterone and sexual function, firstly progesterone plays a role in sexual function in both males and females. It appears that low levels of progesterone may augment sexual function however very high levels seem to always be bad for sexual function. In female mice, progesterone signalling plays a crucial role in facilitating dopamine induced sexual behaviour as mice without the progesterone receptor (PR) show markedly reduced sexual function following a dopamine agonist compared to controls (8). Another study looked into the role of progesterone in androgen dependent sexual behaviour, and found that progesterone plays an important role in sexual behaviour in some male reptiles and rats (9).
Interestingly, progesterone can be linked directly to the dopaminergic system. The striatum is a key component of the dopamine reward system and plays a crucial role in the expression of motivated behaviours (10). Estrogen and progesterone receptors are highly expressed on dopaminergic neurons in the striatum as well as the midbrain and amygdala. A study of female brain activity showed that brain activity changed depending on estrogen and progesterone levels, indicating that brain activity is influenced by the current levels of sex steroids such as estrogen and progesterone. Preclinical data shows that estrogen and progesterone interact to affect neuronal signalling and the effects of estrogen differ in the presence of progesterone (11). Given this, estrogen and progesterone both influence dopaminergic activity in the reward circuitry of the brain. This gives plausibility to the idea that changes in progesterone activity and its receptors could be directly affecting our dopaminergic system, resulting in PAS.
Another study of female mice found that over short time periods of 2-12 hours, progesterone stimulated spontaneous and amphetamine evoked dopamine release and increased tissue concentrations of dopamine in the corpus striatum compared to controls. However, over longer time frames of 24 hours, progesterone appeared to produce an active inhibition with all parameters of dopamine activity with a significant decrease in both spontaneous and amphetamine induced dopamine release compared to controls. In the medial basal hypothalamus, progesterone did not alter spontaneous or marginal amphetamine response but did significantly reduce post super-fusion dopamine concentrations at 24 hours. This study shows that progesterone has a dual effect upon the brain dopaminergic system, it can initially act to facilitate dopamine activity but over sustained, longer time periods it works to inhibit it (12). It should also be noted that there may be sex differences between how progesterone effects dopamine release between males and females.
What is very interesting about this possibility is that high levels of progesterone appear to be capable of significantly reducing sexual function in both females and males in spite of normal levels of either estrogen or testosterone, both of which are known to increase sexual function in females and males respectively. This is significant information, as many have focused on androgen signalling as the root cause of these issues, but this explanation does not help to explain the equally significant effect isotretinoin can have on female sexual function. Though testosterone also plays an important role in female sexual function, it seems unlikely that these effects would affect males and females with equal severity if it related primarily to either testosterone or estrogen specifically, as each hormone plays a significant role in sexual function in either males or females.
Another study indicates the role of progesterone in neural signalling related to the human reward system, which as mentioned before appears to be the core problem in PAS. A study of women found that the menstrual cycle phase and thus levels of gonadal steroid hormones such as estrogen and progesterone influenced dopaminergic reward signalling (13). It has also been shown that dopamine can activate neural progesterone receptors to facilitate reproductive behaviour (14).
Another study of female mice states that multiple signal transduction pathways coexist in the neuroendocrine system for reproductive behaviour. With the progesterone receptor acting as a transcriptional mediator for dopamine as well as progesterone, to achieve integration of neural communication in the central nervous system. This study suggests that dopamine requires the unoccupied progesterone receptor to induce sexual behaviour in mice. Again if PAS does involve high levels of progesterone, this would explain a block of sexual function as progesterone receptors would be occupied or down regulated (15).
Studies have also revealed that retinoic acid appears to be capable of altering progesterone receptor expression in human cells such as human breast cancer cell lines, where exposure to retinoic acid for 24-48 hours after treatment resulted in a significant decrease in the cellular progesterone receptor expression (16). Again the question is whether this also occurs in the brain. Despite this, a lack of progesterone receptor expression could also explain a lack of sexual function if this receptor is indeed required for dopamine signalling as it is in the previous mice study. This info could of course also be completely irrelevant.
Another study found that progesterone decreases mating and sexual activity in male monkeys. It was found that progesterone decreased estradiol uptake in pre-optic area and anterior part of the bed nucleus of stria teminalis by up to 80%. In this study, progesterone decreased sexual activity but not plasma testosterone levels. These findings are also relevant to PSSD as some have suggested a link between progesterone, estrogen and the SERT in PSSD. But this aside, what makes this study interesting is that it shows that estradiol is of great importance to male sexual function and provides a mechanism for how progesterone can affect sexual function without altering testosterone levels (17).
Studies of male mice with progesterone receptor knock outs revealed that mice without the progesterone receptor show enhanced sexual behaviour (this is interesting as it shows that the progesterone receptor may not be required for sexual behaviour). Wild type mice also exhibited greater sexual behaviour when treated with RU486 (progesterone antagonist). What is even more interesting is that mice without the progesterone receptor showed significantly increased androgen receptor expression in the medial pre-optic nucleus and bed nucleus of the stria terminalis. This indicates that PR signalling may work to reduce androgen receptor expression in these areas of the brain (18).
Progesterone and 5AR activity
What is also very interesting to note about progesterone is that progesterone is a 5AR inhibitor. It is thought that when it reaches high concentrations in local tissues, progesterone can significantly inhibit 5AR activity. One study for example, found that progesterone cream applied to the pubic region in males was able to inhibit up to 75% of 5AR activity (19). Another study in human gingival fibroblasts found that progesterone reduced the formation of DHT by 10 fold, where as estrogen increased it by 35% (20). If it is indeed true that isotretinoin greatly increases progesterone levels specifically in the brain, is it possible that this high level of progesterone is enough to significantly inhibit 5AR activity in the brain over a long period of time such as the course of isotretinoin treatment? Additionally, progesterone may also reduce androgen receptor expression in the brain and its 5AR reduced version, 5alpha-dihydroprogesterone (5a-DHP) also antagonises the androgen receptor. Together this seems to provide one of the strongest plausible links between PAS and PFS if these conditions do indeed involve 5AR activity.
Is it possible that one of Isotretinoins’ mechanisms of action is to increase peripheral progesterone levels in the skin by altering the balance of steroidogenic enzymes and thus alter 5 alpha reductase activity?
Moreover, the brain specific production of progesterone from isotretinoin could explain why people with PAS have the same mental side effects as PFS but not as many of the physical side effects such as penile shrinkage and muscle wastage as in PFS. As the 5AR inhibition in PAS is occurring mostly in the brain where as in PFS it occurs across most of the body.
Progesterone, FSH and LH
I have consistently observed PAS sufferers reporting low FSH and LH, especially when considering that some of these individuals are males in their twenties. Studies have shown that administration of progesterone over short periods of time such as 1-6 hours works to increase production of gonadotropin releasing hormone (GnRH) and subsequent production of LH and FSH in pituitary cells. However, when progesterone administration was extended to 12, 36 or 48 hours, there was a significant inhibition of GnRH stimulated FSH and LH release. The results of this study indicate that the anterior pituitary is a major site of action of progesterone in the release of FSH and that 5 alpha reduction of progesterone plays an important role specifically in FSH release. Thus it is possible these low blood results related to FSH and LH, could be indicative of high levels of progesterone signalling in the brain over sustained periods of time (21). However, these observations may also be pure coincidence and not be related to PAS at all. I have also seen examples of people who claim to have PAS who had fairly normal LH levels.
Overall, I think these studies are very interesting as they show a very plausible mechanism of how isotretinoin use could cause sexual dysfunction and many of the related mental side effects. Either through the direct action of either PR signalling on the dopaminergic neurons or a lack of PR signalling through the downrgulation of the PR receptors or through 5AR inhibition.
The other question is whether the changes in gene expression related to progesterone in the brain are Scenario 1: permanent as a result of epigenetics or Scenario 2: temporary changes in response to high concentrations of retinoic acid while on the drug which go back to normal after stopping isotretinoin.
In Scenario 1 if changes in gene expression are permanent, then it is likely that in people with PAS there is a consistently high concentration of progesterone and related compounds in the brain causing persistent sexual dysfunction as well as altering dopaminergic signalling in the reward system etc. causing the majority of other mental symptoms and cognitive issues. In this case, it would make sense to try anti-progesterone substance such as RU-486 (Mifepristone), it could be assumed that by blocking the activity of progesterone, PAS symptoms would improve temporarily while on the drug (this could provide invaluable information). However, as far as I can tell, RU486 would only block the action of progesterone at progesterone receptors, it would not necessarily stop the inhibition of 5AR by progesterone. Thus the use of RU-486 may improve dopaminergic signalling but not improve sexual function. Something to add to this protocol could be DHT such as proviron ( disclaimer: this is only a suggestion, I have no experience with this drug and could equally be very dangerous ) at the same time as RU486 to flood the body with DHT and hope that a sufficient amount seeps into the brain. I have only seen a few reports of PAS people trying RU-486 without much success however I haven’t seen many people try it and its possible the dosages and time spent on the drug were too small and short. Remember we would need a good concentration of RU-486 to reach the brain as there may be very high concentrations of progesterone to notice effects. Let’s call this Scenario 1 protocol . Its also possible that PR signalling is reducing AR expression so AR expression would likely increase when using RU-486. Additionally, it could be worth trying RU-486 with estradiol as I haven’t seen this done before.
In scenario 2 , the development of PAS could very well reflect the development of PFS, as during isotretinoin use there would be a consistent production of progesterone in the brain which would likely block the majority of 5AR activity. Upon finishing treatment, progesterone levels would start to drop and 5AR would come back online causing a PFS like state. In this case, it can only be assumed that protocols which work for PFS would work for PAS. I know there aren’t many but if one arises which works, could be worth trying.
Scenario 3 , it is also possible that changes in progesterone expression were temporary while on the drug. However rather than leading to the same situation as PFS, there is a dysfunction/imbalance in some aspects of progesterone signalling which needs to be corrected. For instance, very high levels of progesterone may have downregulated progesterone receptors so much that there is a lack of negative feedback to reduce progesterone signalling. In this case, we may need to increase progesterone receptor expression through the use of something like estradiol, to recalibrate normal progesterone signalling and regulatory mechanisms. In this case hitting several aspects of progesterone signalling may help flip a switch. Could a scenario 3 protocol of RU-486 with estradiol at the same time make sense? Its also worth noting that in male monkeys, progesterone decreases mating and blocked estradiol uptake in the preoptic area and anterior bed nucleus of the stria terminalis by up to 80%. This study showed that progesterone decreased sexual activity but not plasma testosterone and shows that estradiol is of great importance to male sexual motivation and behaviour.
Summary
Overall I think the relation of progesterone to PAS has not been looked into enough and provides a very plausible mechanism for how PAS and sexual dysfunction arise. Again cases related to ‘Deca Dick’ show how potentially powerful progestogens can be on sexual function and dopamine signalling (mental/cognitive effects) over long periods of time despite normal hormone levels and cessation of the drug. Progesterone also provides a more obvious mechanism for why PAS sexual dysfunction effects males and females to the same severity (this could be a big clue to how PAS arises). It seems like a bit of coincidence that one of the few known hormones which can drastically effect sexual function in both males and females, has also been found to be increased by isotretinoin in the brain.
Progesterone may also help to explain other anomalies (which may or may not be significant) such as:
- PAS people frequently report low levels of FSH and LH in blood tests.
- Self-reported changes in the way dopaminergic drugs such Cannabis, Ritalin and Adderall seem to effect people before and after the drug.
- Why drugs such as L-Dopa which act directly at the dopaminergic system don’t have much, if any effect.
- How 5AR inhibition could occur with isotretinoin despite the drug itself not having much direct activity with the 5AR.
- How potentially normal androgen signalling could be present in PAS but still unable to influence dopamine signalling and sexual function. I find it strange that androgen signalling in my muscles seems to work fine but not in my brain.
I would love to hear any feedback anyone has.
Flynn
Other interesting findings related to progesterone
Progesterone, Estrogen and glutamate/NMDA – Glutamatergic transmission is a key regulator of cognitive domains such as memory and learning and its integration is fundamental for normal cognitive functioning and mental health. Progesterone has been shown to suppress glutamatergic neurotransmission in a dose dependent fashion. Estrogen appears to facilitate glutamate transmission. Progestertone appears to have these effects mainly through non-NMDA receptors but estrogen appears to have these effects through NMDA receptors. To note, estrogen is thought to facilitate dopamine neurotransmission and can also impact the effect of progesterone on this signalling when present. In this case, rats need to be pre-exposed to estrogen and then have progesterone added to stimulate dopamine release (22).
Progesterone, Estrogen and serotonin – Estrogen is thought to be a potent modulator of serotonin activity from the level of serotonin synthesis via the regulation of tryptophan hydroxylase and degradation of 5-HT to the density of 5-HT receptors. Estrogen has been found to increase 5-HT2s mRNA levels in brain areas relevant for the control of mood, mental state and cognition as well as increase serotonin transporter mRNA when administered over long periods of time. However, estrogen treatment has also been observed to decrease mRNA related to serotonergic neurotransmission, for example, 5HT1B autoreceptor mRNa in dorsal raphe and MAO0A mRNA and activity are decreased after estrogen treatment. In cases, acute estrogen administration decreases serotonin transporter mRNA levels and 5HT1A mRNa levels and binding. But the latter effect seems to disappear after a more chronic treatment regimen. Overall it is hard to assign estrogen a definitive status of stimulator or inhibitor of serotonin. Progesterone has been found to affect serotonergic signalling. Chronic progesterone treatment seems to decrease 5HT1A receptor expression in rats, progesterone also decreased 5HT2C receptor expression when it was added to estrogen in macaques (22, 23).
Progesterone and Cannabis - Since isotretinoin I have noticed a marked difference in the effects of cannabis. Prior to Accutane I really enjoyed cannabis and it had dramatically different effects on me. However, since isotretinoin, I no longer enjoy the effects of cannabis and it feels like a completely different drug. A study of mice found that the effects of the main psychoactive compound in cannabis, tetrahydrocannabinol (THC) required both intracellular progesterone receptors and membrane dopamine receptors to initiate effective induction of sexual behaviour in rats (25). This is an interesting link, which could help to explain a link between the change in effect of these drug.
Progresterone and Meth – Another study, found that the up-regulation of progesterone receptors plays a crucial role methamphetamine induced sexual motivation in females (26). This study is interesting as it shows how a drug which acts directly at the dopamine level can be affected by progesterone signalling. I think the effect of drugs which act directly on the dopamine system can provide invaluable clues to PAS as these drugs should technically activate dopamine signalling irrespective of androgen signalling etc. as they don’t interact with androgen receptors. Thus examples of how progesterone signalling can have significant impacts on dopaminergic signalling in response to these drugs shows just how powerful progesterone signalling can be. Despite this, it should also be noted that a lack of androgen signalling could also lead to lower levels of dopamine production, signalling factors and dopamine receptors which may also explain changes in the way dopaminergic drugs take effect.
References
1 - https://forums.t-nation.com/t/feeling-suicidal-because-of-deca-dick/229898
2 - https://pdfs.semanticscholar.org/ac4b/8e66cffb81b27254e96d69a8f12ef3794c9e.pdf
3 - https://forums.t-nation.com/t/9-months-of-deca-dick/197279/106
4 - https://pubmed.ncbi.nlm.nih.gov/3036474/
5 - https://www.ncbi.nlm.nih.gov/pubmed/2373707
6 - https://www.jbc.org/content/265/21/12694.full.pdf
7 - https://pubmed.ncbi.nlm.nih.gov/19744992/
8 - http://citeseerx.ist.psu.edu/viewdo...947B19E?doi=10.1.1.322.7661&rep=rep1&type=pdf
9 - https://www.sciencedirect.com/science/article/abs/pii/S0018506X98914854
11 - http://www.pnas.org/content/104/7/2465.full
12 - https://www.karger.com/Article/PDF/123971
13 - https://www.pnas.org/content/104/7/2465
14 - https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3355960/
15 - https://pubmed.ncbi.nlm.nih.gov/8961281/
16 - https://www.jbc.org/content/265/21/12694.full.pdf
17 - https://www.sciencedirect.com/science/article/abs/pii/S0031938401006060
18 - https://academic.oup.com/endo/article/146/10/4340/2499854
19 - https://books.google.co.uk/books?id=GvDxCAAAQBAJ&pg=PA485&redir_esc=y#v=onepage&q&f=false).
20 - https://onlinelibrary.wiley.com/doi/abs/10.1034/j.1600-051X.1999.t01-4-261101.x
21 - https://pubmed.ncbi.nlm.nih.gov/1616877/
22 - https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4335177/)
23 - https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4335177/)