Thanks for posting, good to see this out.
I am particularly pleased to see Irwig note the essential impossibility of repeated and ultimately pointless calls for placebo controlled study as a useful endeavour due to the huge numbers and time-frames needed given the significant rarity of PFS:
Although a well-designed randomized controlled trial could potentially establish causation, such a trial would probably require over 10,000 subjects in each arm to be adequately powered and would need to last for at least 5 years. It is very unlikely that such a trial would ever be funded or completed.
I recently completed an early draft of a literature review in which I briefly commented on this important point:
Some authors continue to call for repeated placebo controlled trials to determine the existence of persistent effects from finasteride, considering little else to be sufficient (Basaria et al., 2016; Gray & Semla, 2019; Diviccaro et al., 2019). Further study of this kind will not be enlightening nor practically useful to the scientific community, the PFS patient or consumer in the medium term, if ever. It is urgently necessary to acknowledge both the novel nature of the condition and the rarity of the syndrome (Traish 2018) for a pragmatic approach. It is possible that even a large-scale randomized trial would not reach signal. Rarity, however, cannot and should not be construed to justify dismissal of the gravity of this condition (Maksym et al., 2019). This is particularly relevant with consideration to the unpredictability and dose-independence of PFS, and its atypical presentation following withdrawal. Dismissal of retrospective studies is often attempted owing to a perceived lack of credibility in normal instances of ADR. This disease is not a normal ADR, to the point that existing drug reaction algorithms are unable to accommodate it (Healy et al., 2017).
A pragmatic approach to any progress must take the reality of this issue into account, not defer to an arbitrary standard of perceived evidential quality appropriate to a more ordinary adverse drug reaction while patients continue to be driven to suicide by profound and unresolvable suffering. Insistence on the application of a formula that is notedly not fit for purpose in this circumstance manifests as a dereliction of duty. This will be at the expense of lives that could be saved by the most basic of warnings. It is astonishing to consider that mechanistic elucidation may now plausibly precede acknowledgement of a syndrome well reported by patients for two decades. Patients cannot continue to shoulder this global problem in lieu of clinicians.

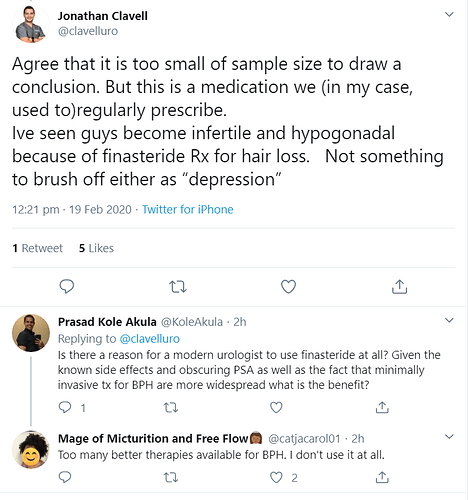

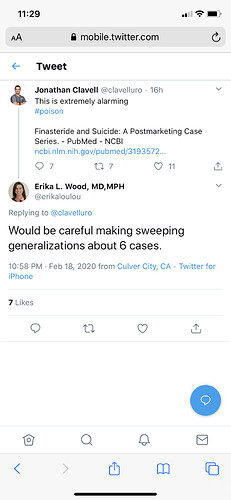
 safe to say we’re in good hands
safe to say we’re in good hands